References
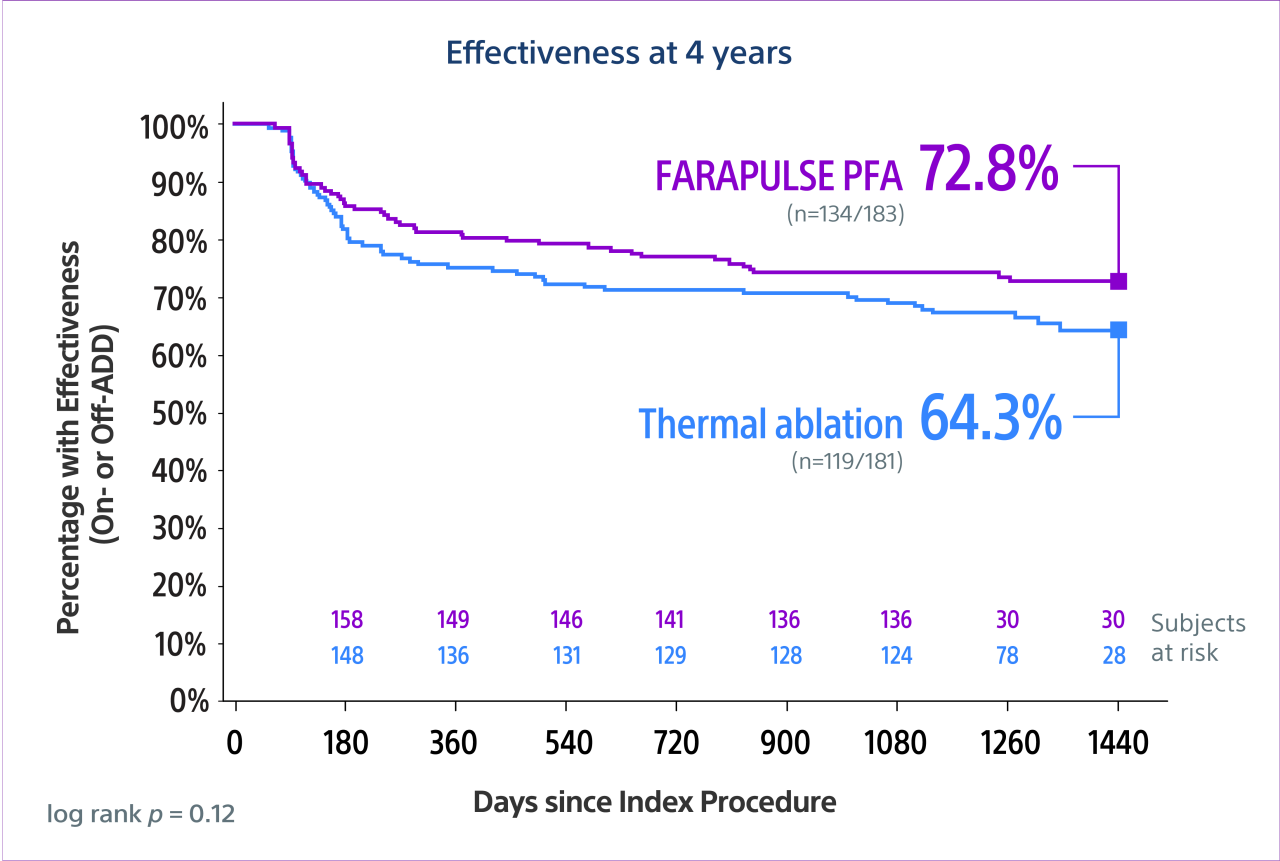
- Reddy, V. Y. (2026). Pulsed field versus conventional thermal ablation for paroxysmal atrial fibrillation: 4-year outcomes in the ADVENT-LTO study. Nature Medicine. https://doi.org/10.1038/s41591-026-04246-4
- Reddy VY, Gerstenfeld EP, Natale A, et al., Pulsed field or conventional thermal ablation for paroxysmal atrial fibrillation. New England Journal of Medicine.2023;Nov2;389(18):1660-1671. doi:10.1056/NEJMoa2307291
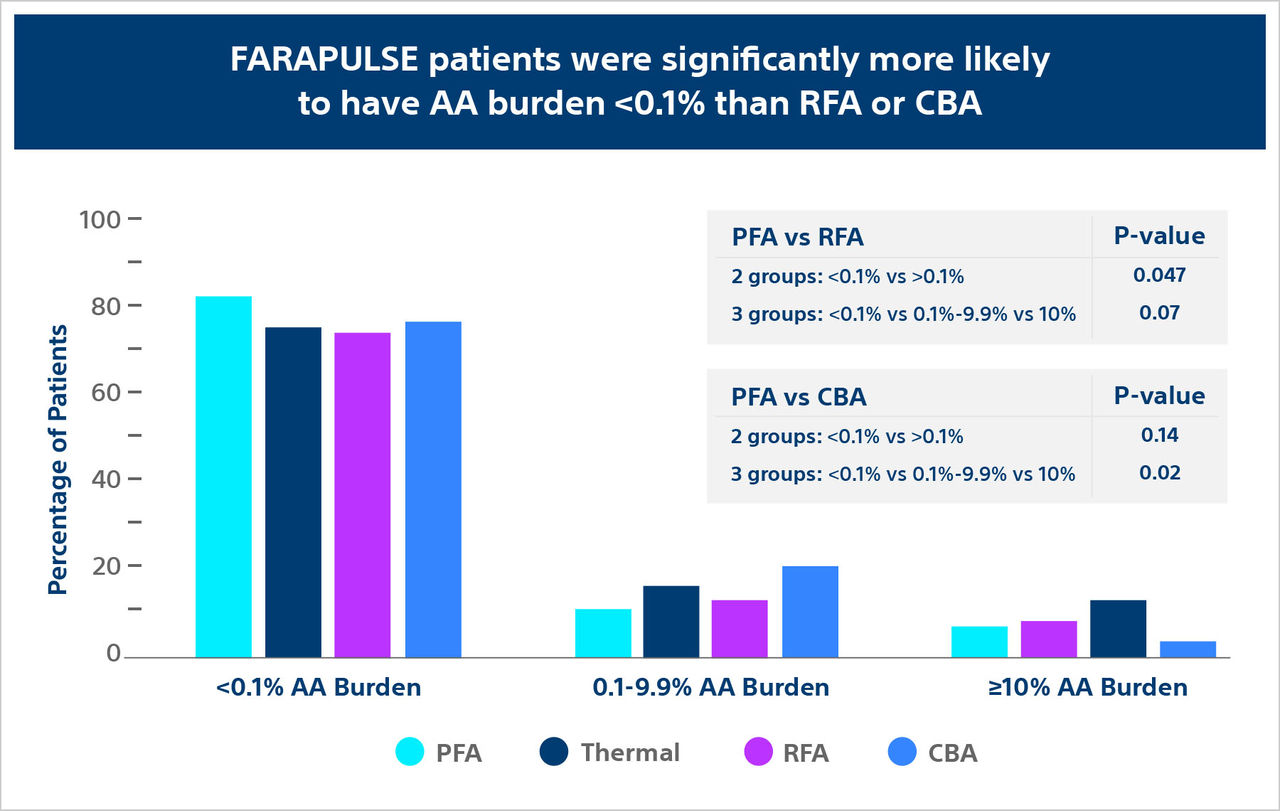
- Reddy V, Mansour M, Calkins H. et al., Pulsed Field vs Conventional Thermal Ablation for Paroxysmal Atrial Fibrillation: Recurrent Atrial Arrhythmia Burden. J Am Coll Cardiol. 2024. 84(1): 61.74. https://doi.org/10.1016/j.jacc.2024.05.001
Boston Scientific Pty Ltd | PO Box 332 Botany NSW 1455 Australia | Tel +61 2 8063 8100
Approved intended use of device may differ for countries with applicable health authority registrations. Always refer to the product labelling for full instructions.
Bench Test or pre-clinical study results may not necessarily be indicative of clinical performance.
Results from clinical studies are not necessarily predicative of results in other cases. Results in other cases may vary.
You may be provided with links to other websites operated by Boston Scientific Corporation (BSC) or other third party websites located outside of Australia & New Zealand. These websites may include information on therapies or products not approved for use in Australia and/or New Zealand and the content may not comply with regulatory requirements in Australia or New Zealand. Please refer to the separate terms of use, privacy policies, and other rules posted on these linked sites before you use them. Third party links are provided merely as a convenience to you, and the inclusion of any link does not imply affiliation or endorsement by BSC.
CAUTION: The law restricts these devices to sale by or on the order of a physician.
Indications, contraindications, warnings, and instructions for use can be found in the product labelling supplied with each device or at www.IFU-BSCI.com.
Products shown for INFORMATION purposes only and may not be approved or for sale in certain countries.
©2023 Boston Scientific Corporation or its affiliates. All rights reserved.
EP-1678203-AA